[box type=”bio”] Learning Point of the Article: [/box]
Conversion total hip arthroplasty can be completed safely and with positive outcomes in patients with factor XI deficiency.
Case Report | Volume 10 | Issue 3 | JOCR May – June 2020 | Page 80-84 | Joseph R. Young, Jared Roberts. DOI: 10.13107/jocr.2020.v10.i03.1760
Authors: Joseph R. Young[1], Jared Roberts[1]
[1]Department of Orthopedic Surgery, Albany Medical Center, Albany, New York, USA.
Address of Correspondence:
Dr. Joseph R. Young,
43 New Scotland Ave, MC-184, Albany – 12208, New York.
E-mail: joseph.russell.young@gmail.com
Abstract
Introduction: Factor XI (FXI) deficiency is a hematologic condition that is rarely encountered by the arthroplasty surgeon. Effective risk assessment and perioperative management are paramount in minimizing the risk of intra- and post-operative bleeding in this patient population. An interdisciplinary approach is crucial in minimizing complications and achieving successful outcomes. We present the case of a patient that successfully underwent conversion total hip arthroplasty (THA) following failed internal fixation of a proximal femur fracture.
Case Report: A 71-year-old man with a history of FXI deficiency presented with significant right-sided hip pain secondary to post-traumatic arthritis from a previously treated right proximal femur fracture. The patient underwent removal of the cephalomedullary nail and conversion to a THA. Before the procedure, a comprehensive perioperative plan was enacted to manage the patient’s FXI deficiency. The patient underwent several infusions of aminocaproic acid and tranexamic acid (TXA) in an effort to prevent intra- and post-operative bleeding. The surgery was completed with excellent hemostasis and no post-operative complications.
Conclusion: Patients with FXI deficiency can successfully undergo conversion THA surgery; however, an individualized hematologic plan must be enacted to minimize complications and maximize surgical outcomes and patient satisfaction. This case demonstrates that the antifibrinolytic agents, aminocaproic acid and TXA, can be successfully used for hematologic prophylaxis in the perioperative period for this population of patients.
Keywords: Arthroplasty, Conversion, Deficiency, Factor XI, Hemophilia, Total
Introduction
Surgical management of patients with factor XI (FXI) deficiency, also known as hemophilia C, presents unique challenges to the orthopedic arthroplasty surgeon. Significant pre-operative planning is necessary to mitigate the risk of bleeding or other complications faced by these patients throughout the perioperative period. Through an interdisciplinary approach, such patients can be successfully managed through joint replacement surgery with the expectation of a successful outcome. We report on a case of a 71-year-old man with a history of FXI deficiency who successfully underwent conversion total hip arthroplasty (THA) following failed internal fixation of a proximal femur fracture. To the best of our knowledge, there have been no previous reports on the management of patients with FXI deficiency undergoing this procedure. The patient provided informed consent for the publication of the details of this case.
Case Report
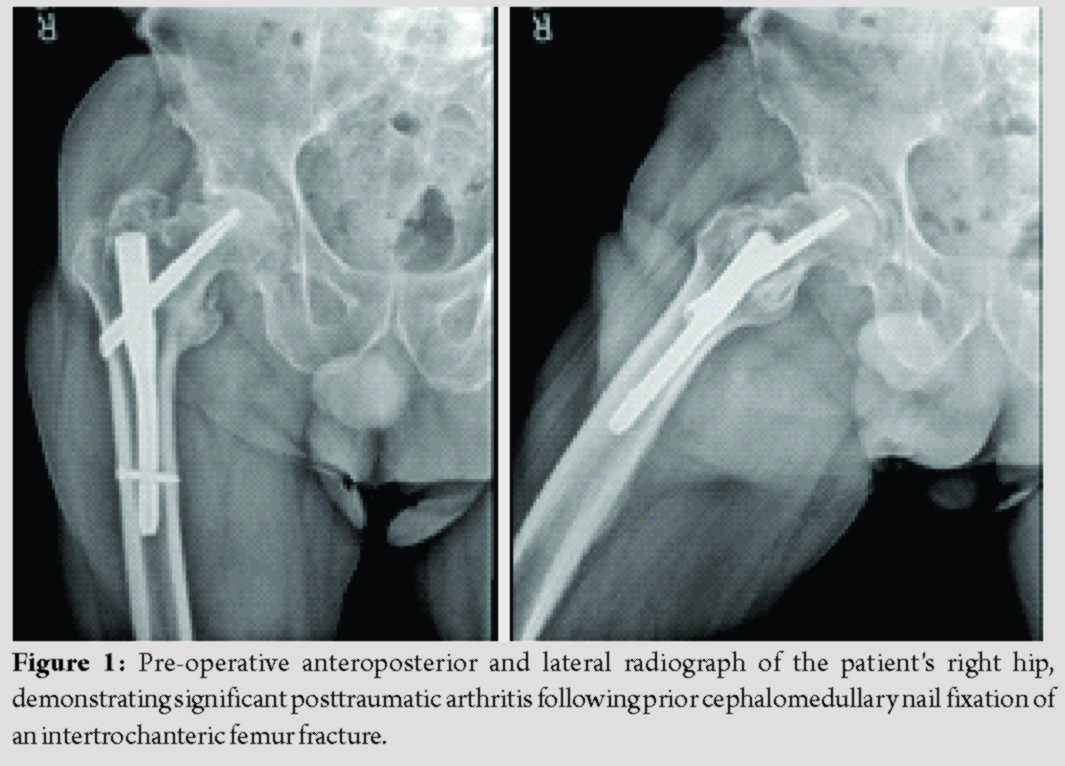
A 71-year-old man with a history of FXI deficiency presented to the orthopedic clinic with complaints of progressively worsening pain in his right hip. Of note, the patient had previously undergone cephalomedullary nail fixation of a right proximal femur fracture 3 years prior by another local orthopedic surgeon. The patient subsequently went onto heal this fracture; however, he did develop severe degenerative changes in his right hip along with significant post-traumatic arthritis (Fig. 1). Surgical options along with their risks and benefits were reviewed with the patient who ultimately elected to undergo conversion THA. Of note, the patient had a history of significant post-operative bleeding following a right-sided hernia repair done 19 years prior. At that time, his FXI deficiency was treated with only a single dose of fresh frozen plasma (FFP). Since then, he had undergone cephalomedullary nail fixation of both his left and right hips 3 and 5 years prior, respectively. He did not experience any post-operative bleeding or thromboembolic complications following either hip fracture surgery. The patient’s hematologist devised the following perioperative management strategy before his first hip fracture surgery: Preoperatively, a 5-g IV bolus of aminocaproic acid would be administered followed by aminocaproic acid as continuous venous infusion of 5 g in a 250 mL solution at 50 mL/h throughout the perioperative period. Postoperatively, tranexamic acid (TXA) would be administered as a 1 g bolus followed by 1 g every 8 h for 72 h for a total of nine doses. The patient would then be discharged on oral TXA at 1300 mg every 8 h for the first 7 days, with the frequency decreased to every 12 h for the subsequent week. He would also be administered subcutaneous enoxaparin 40 mg daily for venous thromboembolism prophylaxis.
Before conversion THA surgery, the patient was seen in the hematology clinic so that his perioperative plan could be reviewed before this surgery. The patient had been previously seen in this clinic at the time of this right hip fracture. At that time, the patient’s FXI deficiency was confirmed, with his factor XI assay at 1.9%, well below the normal value of 50–150% activity. Given the patient’s success with the aforementioned hematologic plan, it was agreed that the same protocol would be used for his THA conversion surgery. The patient was taken to the operating room for removal of the cephalomedullary nail and subsequent conversion to a THA. Pre-operative hemoglobin and hematocrit were 12.4 g/dL and 35.6%, respectively. Using the previous surgical incision, removal of the cephalomedullary nail was completed with some difficulty as the distal locking screw was stripped at the time of removal. Once this was completed, attention was turned to completing the THA. Utilizing a standard anterior approach, the proximal femur was cut in appropriate position, the acetabulum was reamed up to a size 57, and a size 58 cup was press-fit with excellent purchase. The cup was fixed with two screws into the ilium. Next, the femur was prepared. An 8″, size 18 Solution (Depuy, Warsaw Indiana) stem with a 36 + 5 head was deemed to provide adequate stability, and the final components were implanted. Blood loss was estimated at 700 mL. The patient was made toe-touch weight-bearing to the right lower extremity. There were no intraoperative complications experienced.
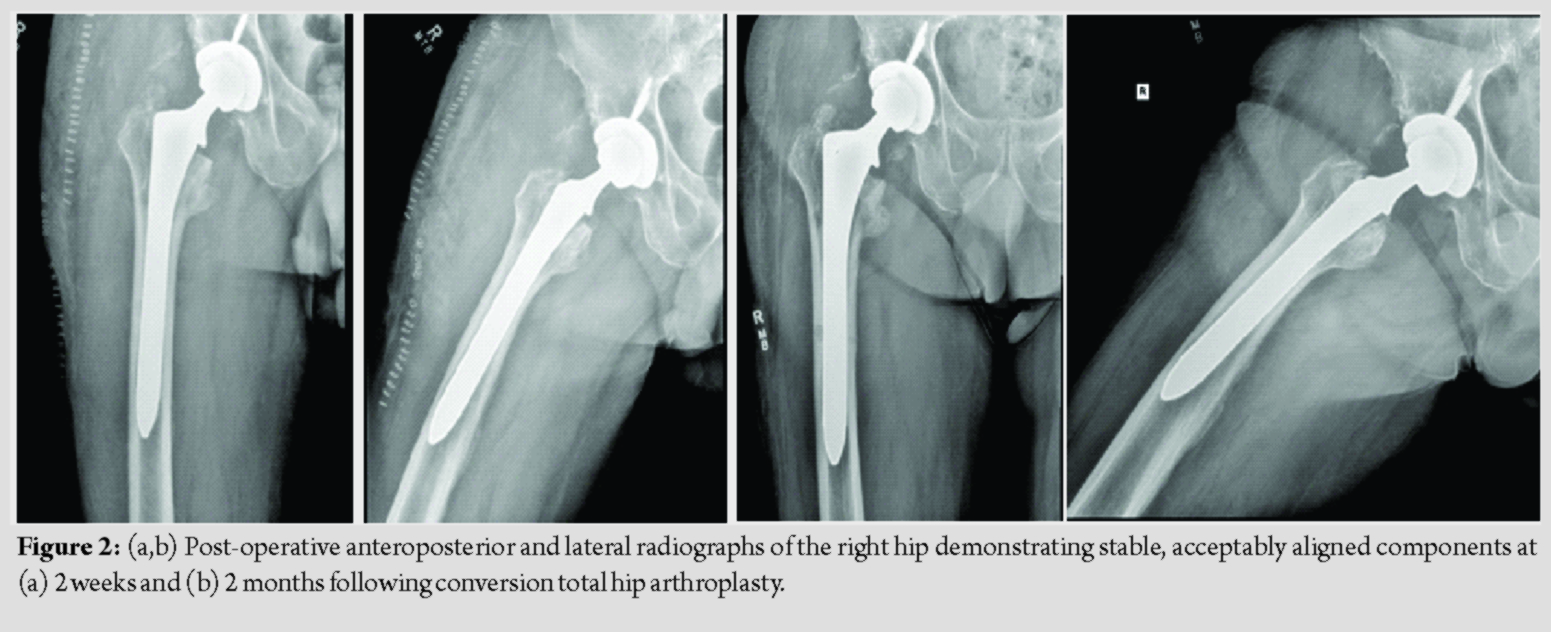
The patient was placed on the pre-established protocol during and after the surgery. He was cleared by physical therapy on post-operative day 1, and plans were made for discharge to home with a rolling walker. The patient was observed closely for signs of bleeding, and he remained asymptomatic throughout his hospital course. The patient’s hemoglobin and hematocrit did decrease to 8.0 g/dL and 24.6% before stabilizing, and no transfusion was necessary. Ultimately, the patient was discharged to home in stable condition on post-operative day 3, shortly after finishing nine doses of IV TXA per the previously established protocol. Follow-up was obtained at 2 weeks, 2 months, 5 months, and 10 months, with post-operative radiographs obtained at 2 weeks and 2 months (Fig. 2). The patient continued to progress to weight-bearing as tolerated without issue. Follow-up X-rays demonstrated stable and well-aligned components. His pre-operative pain improved dramatically. Despite some mild stiffness and a small leg length discrepancy, the patient’s overall clinical status improved significantly and he remains very satisfied with the outcome of his surgery. He did not have any bleeding-related complications throughout his perioperative course. At 10 months, the patient reported no complications, and he noted that he was back to full activity including hiking and snowshoeing without pain or other noticeable symptoms.
Discussion
Factor XI deficiency, also known as hemophilia C, is a rare autosomal recessive bleeding disorder with an incidence of approximately 1 in 1 million people worldwide [1]. First described in the 1950’s, the condition is especially prevalent in the Ashkenazi Jewish population, with a frequency of approximately 8% [1, 2]. In contrast to the more severe hemophilia A and B, the condition is milder, is more likely to involve both genders, and is not associated with spontaneous hemorrhage into joints or soft tissues [2]. Historically, this condition was detected after a significant bleeding event following trauma or surgery; however, at present, it is more likely to be diagnosed incidentally following a prolonged aPTT on routine pre-operative evaluation [3]. Patients with FXI deficiency present unique management challenges in the perioperative period. The bleeding phenotype is highly variable, and it can be difficult to predict one’s bleeding risk during and after surgery [4]. A comprehensive pre-operative history and physical examination must be completed for risk stratification and to assess the need for prophylactic hemostatic intervention [4]. In our case, prophylactic management was necessary given the patient’s prior bleeding episode following surgery, as well as the complex nature of the operation and potential for significant intraoperative blood loss. There is currently no standardized prophylactic treatment regimen established for patients with FXI deficiency undergoing major surgery, and no optimal tool exists to predict the bleeding risk in these patients [5]. Multiple prophylactic treatment modalities have been utilized. These include FFP or thawed plasma (TP) transfusion, recombinant activated factor VII, FXI concentrate, vasopressin (DDAVP), and antifibrinolytic agents such as aminocaproic acid or TXA [4]. Ultimately, the type of prophylaxis should be individually tailored to each patient, taking into account, the patient’s bleeding history, as well as the fibrinolytic activity of the tissue at the operative site and the type of surgery planned. Many authors advocate the use of prophylactic replacement therapy using FFP or FXI concentrate [3, 6, 7, 8]. FFP has proven to be effective, however, risks do exist for transfusion-associated circulatory overload (TACO) and infection transmission given the significant amount of volume needed to achieve desired levels [4, 9]. FXI concentrate minimizes these risks but portends its own increased risk of thrombosis in approximately 10% of cases [3]. In addition, this therapy has limited availability in the United States [6]. The previous cases have been reported in patients with FXI deficiency undergoing primary THA. Santoro et al. demonstrated the use of factor XI concentrate in a 58-year-old male undergoing THA who had previously had a severe allergic reaction to FFP. The investigators administered factor XI concentrate transfusions throughout the perioperative period with excellent hemostasis and no reported complications [6]. Sano et al. reported on a 77-year-old woman who underwent THA successfully with perioperative FFP transfusions and noted no hemostatic complications [7]. Our patient did not wish to receive FFP, as he had experienced significant post-operative bleeding years earlier when subtherapeutic levels were administered during a hernia repair. In cases such as ours, antifibrinolytic agents can be utilized to counteract the profibrinolytic effect of the disease [10]. While reports on the use of aminocaproic acid and TXA used as prophylaxis in orthopedic patients with FXI deficiency are limited, these agents have been used with some frequency in oral surgery patients [11]. A Cochrane Review in 2015 demonstrated that low-level evidence exists that suggests the use of aminocaproic acid and TXA is associated with a reduction in blood loss, number of bleeding events, and the need for clotting factor concentrates in patients with hemophilia undergoing minor oral surgery or dental extractions [12]. A similar treatment strategy proved to be successful in our case.
Conclusion
Perioperative management of patients with FXI deficiency presents a distinct challenge to the orthopedic arthroplasty surgeon. A multidisciplinary approach with a hematologist must be utilized to ensure that a comprehensive plan for perioperative management is in place before surgery. Effective hematologic management is crucial in minimizing the patient’s risk of post-operative bleeding and its associated sequelae. While there are multiple prophylactic treatment modalities that can be used, our case illustrates that a successful outcome can be achieved through perioperative management with antifibrinolytic therapy alone.
Clinical Message
Conversion THA following failed hip fracture fixation can be completed safely and effectively in patients with factor XI deficiency provided that a comprehensive perioperative plan is in place to minimize the risk on intra- and post-operative bleeding.
References
1. Franchini M, Veneri D, Lippi G. Inherited factor XI deficiency: A concise review. Hematology 2006;11:307-9.
2. Gomez K, Bolton-Maggs P. Factor XI deficiency. Haemophilia 2008;14:1183-9.
3. Salomon O, Seligsohn U. New observations on factor XI deficiency. Haemophilia 2004;10 Suppl 4:184-7.
4. Alsammak MS, Ashrani AA, Winters JL, Pruthi RK. Therapeutic plasma exchange for perioperative management of patients with congenital factor XI deficiency. J Clin Apher 2017;32:429-36.
5. Salomon O, Budnik I, Avishai E, Tamarin I, Bashari D, Dardik R, et al. Single low dose of rFVIIa combined with antifibrinolytic agent is a simple and safe treatment for factor XI-deficient patients undergoing surgery. Thromb Haemost 2019;119:1927-32.
6. Santoro C, Goldberg I, Bridey F, Figgie MP, Karila-Israel D, Haviland K, et al. Successful hip arthroplasty in an adult male with severe factor XI deficiency using hemoleven®, a factor XI concentrate. Haemophilia 2011;17:777-82.
7. Sano K, Homma Y, Baba T, Ando J, Matsumoto M, Kobayashi H, et al. Total hip arthroplasty via the direct anterior approach with Kerboull-type acetabular reinforcement device for an elderly female with factor XI deficiency. SICOT J 2017;3:11.
8. Wheeler AP, Gailani D. Why factor XI deficiency is a clinical concern. Expert Rev Hematol 2016;9:629-37.
9. Bolton-Maggs PH, Perry DJ, Chalmers EA, Parapia LA, Wilde JT, Williams MD, et al. The rare coagulation disorders–review with guidelines for management from the United Kingdom haemophilia centre doctors’ organisation. Haemophilia 2004;10:593-628.
10. Livnat T, Tamarin I, Mor Y, Winckler H, Horowitz Z, Korianski Y, et al. Recombinant activated factor VII and tranexamic acid are haemostatically effective during major surgery in factor XI-deficient patients with inhibitor antibodies. Thromb Haemost 2009;102:487-92.
11. McCarthy ML, Ordway SM, Jones RM, Perkins JG. Successful perioperative management in a patient with factor XI deficiency. BMJ Case Rep 2018;2018:bcr2017222434.
12. Van Galen KP, Engelen ET, Mauser-Bunschoten EP, Van Es RJ, Schutgens RE. Antifibrinolytic therapy for preventing oral bleeding in patients with haemophilia or Von Willebrand disease undergoing minor oral surgery or dental extractions. Cochrane Database Syst Rev 2015;12:CD011385.
 |
 |
| Dr. Joseph R. Young | Dr. Jared Roberts |
| How to Cite This Article: Young JR, Roberts J. Conversion Total Hip Arthroplasty Following Failed Hip Fracture Fixation in a Patient with Factor XI Deficiency: A Case Report. Journal of Orthopaedic Case Reports 2020 May-June;10(3): 80-84. |
[Full Text HTML] [Full Text PDF] [XML]
[rate_this_page]
Dear Reader, We are very excited about New Features in JOCR. Please do let us know what you think by Clicking on the Sliding “Feedback Form” button on the <<< left of the page or sending a mail to us at editor.jocr@gmail.com