[box type=”bio”] Learning Point of the Article: [/box]
Late presentation of symptomatic amputation neuroma is an unusual condition, mostly due to cutting the nerve without adequate pull in the previous surgery which requires early detection and surgical treatment at the index procedure to gain better functional outcomes.
Case Report | Volume 10 | Issue 6 | JOCR September 2020 | Page 28-31 | Puripun Jirangkul, Arkaphat Kosiyatrakul, Pawin Gajaseni. DOI: 10.13107/jocr.2020.v10.i06.1862
Authors: Puripun Jirangkul[1], Arkaphat Kosiyatrakul[1], Pawin Gajaseni[1]
[1]Department of Orthopaedics, Phramongkutklao Hospital and College of Medicine, Bangkok, Thailand.
Address of Correspondence:
Dr. Puripun Jirangkul,
Department of Orthopaedics, Phramongkutklao Hospital and College of Medicine, Bangkok, Thailand.
E-mail: dr.puripun@gmail.com
Abstract
Introduction: Post-traumatic amputation neuroma is one of the common complications that involve optimal functional outcomes. The current literature has a few examples of late presentation of traumatic stump neuroma. However, no available examples of late presentation of recurrent symptomatic amputation neuroma causing impaired functional outcomes have been reported.
Case Report: We present a case of recurrent symptomatic stump neuroma after above-knee amputation and neurectomy for 28 years.
Conclusion: Late presentation of recurrent stump neuroma is an unusual condition, requiring early detection and treatment to gain better functionality. The patient scenario is unique and valuable for future management, including case awareness regarding this unique pathology.
Keywords: Amputation neuroma, symptomatic neuroma, recurrent neuroma.
Introduction
Amputation is a common procedure resulting in loss of a limb, usually from injury or disease. It involves preserving life, preventing the infection spreading, promoting mobility, and alleviating pain. Advances in new prosthetics for the foot and knee have enabled amputees to pursue high activity levels with diverse options and better functionality. However, many post-amputation complications were described in several related studies. Pain is the most common and inevitable consequence of amputation. Several types of unpleasant sensations and pain following an amputation include residual limb pain(RLP), phantom limb pain(PLP), phantom limb sensation(PLS), and symptomatic neuroma. One long-term study of patients with transfemoral amputations reported that 64.5% of patients experienced stump pain [1] compared with 21% reported by another study [2]. Post-amputation pain can be isolated to RLP and PLP. Pain not only results from tissue trauma during the surgical procedure but also from nerve injuries. Normally, pain from post-amputation neuroma occurs within a year of surgery that should be distinguished from other causes of stump pain [3, 4].
Case reports of symptomatic neuroma resulting in neuropathic stump pain have been published. Most presented within a year after amputation, only a few case reports have revealed late presentation [5, 6]. To the best of our knowledge, this is the first case of late presentation of recurrent symptomatic amputation neuroma. We reviewed the literature and discussed the management. This report illustrates importance of early detection and surgical treatment at the index procedure to gain better functional outcomes and prevent post-amputation neuroma.
Case Report
A 67-year-old male veteran presented a 28-year history of above the left knee amputation because of blast injury. He was a patient with well controlled type II diabetic mellitus attending our clinic with above the knee amputation stump pain and could not wear his prosthesis for 3 weeks. The history report from rehabilitation department was that the patient experienced a blast injury on his left thigh and leg. He underwent above the knee amputation 28 years ago. Approximately 6 weeks after the surgery, the prosthesis was made for the patient and a rehabilitation at home program was started. Neuropathic stump pain was recorded 3 months thereafter. Stump neuroma and bone spur were suspected to be the pain origin. The patient underwent stump revision with excision of neuroma and bone spur. After the second operation, no evidence was found of stump complication, and the patient was admitted in the military ward with orthopedic trauma services. He had some degree of posttraumatic stress disorder countered with psychiatric team support. No suicidal or homicidal ideation while admitted was noted. Daily life activities and self-care ability were supported by a physiotherapist. The patient was discharged at every 2–3 weeks follow-up appointments for 2 months and every 3 months thereafter. After 12 months, he missed his scheduled follow-up appointment.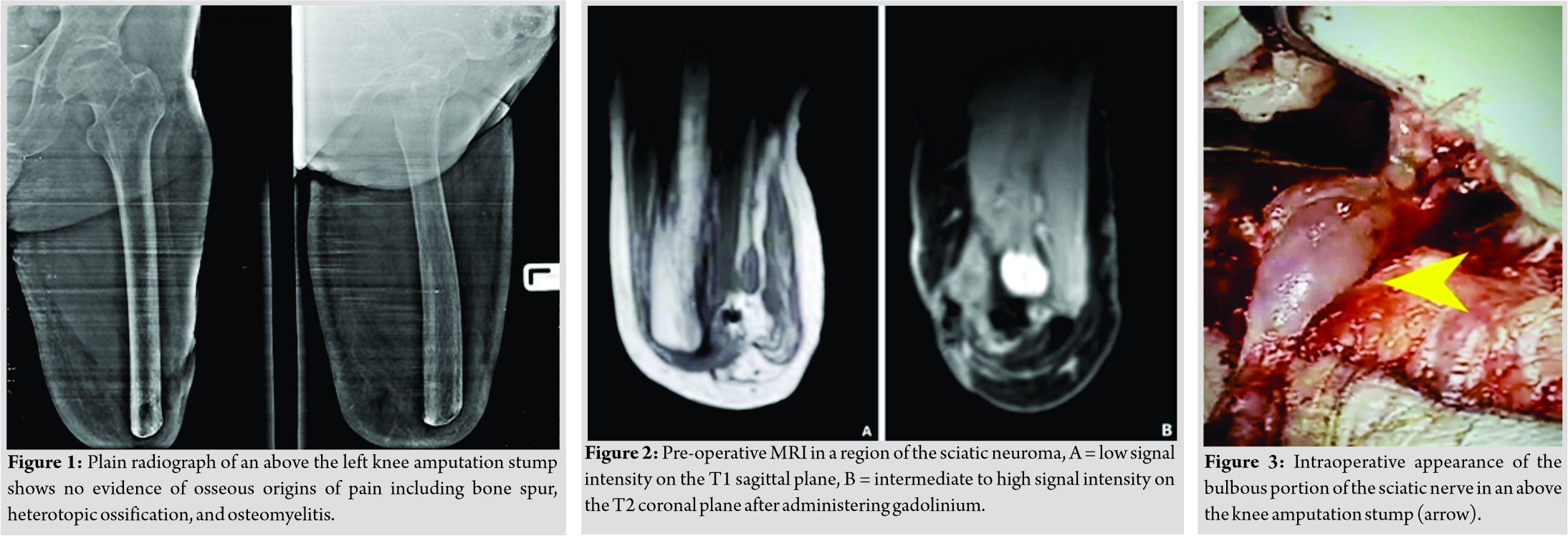
Twenty-eight years later, the patient attended our clinic presenting severe burning and stabbing sensation and clinical sign suggesting sciatic neuroma. He exhibited limited ability to use his prosthesis and subsequently stopped wearing the prosthesis because of uncontrolled pain for 3 weeks. He complained his prosthesis had not fit the stump for 1 year because of muscle atrophy and mild intensity of the stump pain for 9 months. However, he encountered to keep on tolerating stump pain and finally presenting severe neuropathic pain. The physical examination revealed positive Tinel’s sign and palpable lump at the proximity of the posterior stump. The diagnosis block test result with lidocaine was positive. Conventional radiography (Fig. 1) showed no evidence of osseous origins of pain such as heterotopic ossification or osteomyelitis. Magnetic resonance imaging (MRI) (Fig. 2a and b) showed features suggesting a sciatic neuroma. The initial treatment was non-operative including nonsteroidal anti-inflammatory drugs, high-dose pregabalin, physical therapy, local steroid injection, and high-power laser therapy. A new prosthesis was made for the patient. At a routine follow-up clinic visit, the patient still limited his daily life activities due to neuropathic stump pain. The decision was made to perform sciatic nerve traction neurectomy and muscle implantation, which are techniques involving gentle pulling of the bulbous portion of the nerve before proximal dissection allowing the tension to retract the sciatic nerve back up into the soft tissue and bury the end of the proximal nerve stump within the surrounding muscle [7, 8, 9]. This theoretically reduces pressure on the sciatic nerve ending and prevents neuroma formation. Intraoperative appearance of sciatic neuroma at the above-knee amputation stump is shown in (Fig. 3). After approximately 30 minute of neurectomy and muscle implantation, the stump was revised and closed. The patient received in a soft dressing about the wound and multimodal analgesia was administrated. After the operation, no surgical wound infection, PLS or PLP, was observed. The patient was able to wear the prosthesis with optimally functional outcome and proper physiotherapy protocol after surgical wound stitch off. At the first follow-up appointment, the patient was asymptomatic. At the most recent follow-up visit, 3 years after the surgery, he continued to perform daily life activities well, with no clinical signs of unpleasant sensation or recurrence.
Discussion
Post-amputation pain is highly prevalent after limb amputation but remains an extremely challenging pain condition to treat. The incidence and prevalence of amputation stump pain vary [1, 2]. Several authors have studied the causes and pathology of post-amputation pain. Post-amputation pain can be isolated to the RLP or can occur as PLP. PLP should be distinguished from RLP, also known as “stump” pain, which is localized to the remaining body part after amputation. In the post-operative and early stages of rehabilitation, the challenge is to distinguish RLP between nociceptive and neuropathic causes. Pain not only results from tissue trauma during the surgical procedure but also from nerve injuries. Normally, pain from post-amputation neuroma occurs within a year of surgery [3, 4]. Their pathologies can be described as the bulbous tumor of a nerve ending that occurs following traumatic transection related to nerve damage. Wallerian degeneration occurs distally and when regrowth occurs, the axon fibers and new Schwann cells grow in a disorganized manner. The clinical findings are often non-specific; however, further investigation such as MRI is useful to detect these complications early. Neuroablative, non-neuroablativetherapies [10, 11] and surgical procedures including traction neurectomy with or without implantation of the proximal nerve stump into adjacent tissues [7, 12] and nerve-to-nerve anastomosis [7, 13] are common procedures performed to relieve neuropathic pain. In this case, conservative treatments were administrated unsuccessfully for 3 months. Therefore, the patient needed surgical treatment. According to the literature review, traction neurectomy alone appears to produce a high rate of symptomatic recurrent stump neuroma [7].On the other hand, good to excellent results were demonstrated in 82% of patients from neuroma excision and implantation of the nerve end within muscle [14]. Additional surrounding tissue implantation techniques were described including within the bone [15]or veins [16]; these techniques have not been found to meet universal success [7]. Our decisions were neurectomy and muscle implantation, which constitute commonly performed techniques for terminal neuromas and provide good to excellent outcomes. Our patient, with late presenting stump pain, represents an example of late recurrent symptomatic sciatic neuroma. The unexpected presentation of recurrent neuropathic stump pain after an extremely long pain-free interval surprised all clinicians. According to the literature reviewed, the most recent publications reported symptomatic neuroma occurring within a year of amputation and can continue to enlarge for 3 years [2, 3, 17]. On the contrary, the onset of recurrent or persistent neuroma-type pain was at a mean follow-up of 37 months [12](range, 11–91 months). The case of late presenting recurrent symptomatic neuroma more than 91 months after limb amputation has not been reported in the literature. However, this patient differs from those in related studies in at least two important aspects. First, it might be possible that a recurrent stump neuroma occurred within a couple years of previous neurectomy and stump revision rather than a late neuroma formation. However, the specific signs and symptoms of sciatic neuroma occurred 28 years post-amputation. Radiographic imaging investigation confirmed this diagnosis. Second, this case is quite unique in that our patient, therefore, had no stump complications including PLS, PLP during, and leading normal daily life activities with his prosthesis until 28 years later. The onset of symptoms suggesting recurrent amputation sciatic neuroma was 28 years. Furthermore, this case failed conservative treatment with physical therapy, nonsteroidal antiinflammatory drugs, high-dose pregabalin, local steroid injection, and high-power laser therapy. Quite possibly, patients with longer amputation duration might not respond well to non-operativetreatment [8]. The treatment goal of late presenting neuroma is relieving pain and gaining functionality, which can often be achieved by surgical procedure. The late presentation of recurrent posttraumatic amputation neuroma is an uncommon condition rarely encountered and was probably caused by improper previous surgical technique. The cause of stump pain was recurrent sciatic neuroma, mostly due to cutting the nerve without adequate pull in the previous surgery permitting spontaneous retraction into the muscles. Furthermore, disused muscle atrophy and skin breakdown, by retracting scar tissue, were possible causes of the delayed presentation of neuropathic stump pain, and this clinical scenario has not been reported previously. The example is valuable for future management and case awareness regarding this unique pathology. Further study should clarify the exact pathophysiology resulting in late recurrent presenting of symptomatic post-amputation neuroma.
Conclusion
The late presentation of post-amputation neuroma is rare. Clinical awareness and early detection and treatment are of paramount importance to gain better functionality. Appropriate treatment should be administered after clinical signs and symptoms suggestive of stump neuroma were detected. Radiographic imaging such as ultrasonography and MRI may be unnecessary to confirm diagnosis. Conservative treatment should not take too long because long amputation duration and recurrent cases might not respond well. This may delay appropriate surgical treatment and worsen results.
Clinical Message
As the cause of late presentation of stump pain was recurrent sciatic neuroma, mostly due to cutting the nerve without adequate pull in the previous surgery permitting spontaneous retraction into muscles. It is important to recognize the appropriate amputation technique. Although rare, late presentation of neuropathic stump pain is probably caused by contraction of surrounding scar tissue and disused muscle atrophy. Early treatment is essential to gain better functional outcome.
References
1. Ebrahimzadeh MH, Fattahi AS. Long-term clinical outcomes of Iranian veterans with unilateral transfemoral amputation. Disabil Rehabil 2009;31:1873-7.
2. Jensen TS, Krebs B, Nielsen J,Rasmussen P. Immediate and long-term phantom limb pain in amputees: Incidence, clinical characteristics and relationship to pre-amputation limb pain. Pain 1985;21:267-78.
3. Dougherty PJ. Long-term follow-up of unilateral transfemoral amputees from the Vietnam War. J Trauma 2003;54:718-23.
4. Necmioglu S, Subasi M, Kayikci C, Young DB. Lower-limb landmine injuries. Prosthet Orthot Int 2004;28:37-43.
5. Kitcat M, Hunter JE, Malata CM. Sciatic neuroma presenting forty years after above-knee amputation. Open Orthop J 2009;3:125-7.
6. Daniilidis K, Stukenborg-Colsman CM, Ettinger M, Windhagen H. Huge sciatic neuroma presented 40 years after traumatic above knee amputation. Technol Health Care 2013;21:261-4.
7. Eberlin KR, Ducic I. Surgical algorithm for neuroma management: A changing treatment paradigm. Plast Reconstr Surg Glob Open 2018;6:e1952.
8. Gorkisch K, Boese-Landgraf J, Vaubel E. Treatment and prevention of amputation neuromas in hand surgery. Plast Reconstr Surg 1984;73:293-9.
9. Smith DG, Fergason JR. Transtibial amputations. Clin Orthop 1999;361:108-15.
10. West M, Wu H. Pulsed radiofrequency ablation for residual and phantom limb pain: A case series. Pain Pract 2010;10:485-91.
11. Gruber H, Kovacs P, Peer S, Frischhut B, Bodner G. Sonographically guided phenol injection in painful stump neuroma. AJR Am J Roentgenol 2004;182:952-4.
12. Pet MA, Ko JH, Friedly JL, Smith DG. Traction neurectomy for treatment of painful residual limb neuroma in lower extremity amputees. J Orthop Trauma 2015;29:e321-5.
13. Barberá J, Albert-Pampló R. Centrocentral anastomosis of the proximal nerve stump in the treatment of painful amputation neuromas of major nerves. J Neurosurg 1993;79:331-4.
14. Dellon AL, Mackinnon SE. Treatment of the painful neuroma by neuroma resection and muscle implantation. Plast Reconstr Surg 1986;77:427-38.
15. Mass DP, Ciano MC, Tortosa R, Newmeyer WL, Kilgore ES Jr. Treatment of painful hand neuromas by their transfer into bone. Plast Reconstr Surg 1984;74:182-5.
16. Herbert TJ, Filan SL. Vein implantation for treatment of painful cutaneous neuromas. A preliminary report. J Hand Surg Br 1998;23:220-4.
17. Sehirlioglu A, Ozturk C, Yazicioglu K, Tugcu I, Yilmaz B, Goktepe AS. Painful neuroma requiring surgical excision after lower limb amputation caused by landmine explosions. Int Orthop 2009;33:533-6.
 |
 |
 |
| Dr. Puripun Jirangkul | Dr. Arkaphat Kosiyatrakul | Dr. Pawin Gajaseni |
| How to Cite This Article: Jirangkul P, Kosiyatrakul A, Gajaseni P. Late Presentation of Recurrent Symptomatic Amputation Neuroma. Journal of Orthopaedic Case Reports 2020 September;10(6): 28-31. |
[Full Text HTML] [Full Text PDF] [XML]
[rate_this_page]
Dear Reader, We are very excited about New Features in JOCR. Please do let us know what you think by Clicking on the Sliding “Feedback Form” button on the <<< left of the page or sending a mail to us at editor.jocr@gmail.com




